
So, such as many other tools in the QMS, the internal audit checklist is a time-saving tool that will benefit to prevent mistakes, and if you are interested in implementing a lean-but-useful QMS, then tools like the internal audit checklist are invaluable to help you in this endeavour. When you have completed an internal audit, you do not want to search that you have omit to collect the proper information and require re-scheduling your audit to complete it. The checklist is best used by trained and practicing auditors to evaluate or assess Quality Management Systems requirements based on the standard as you. When you are discussing your process plans, you can write down what you require to check, and in this way you can ensure that nothing important is forgotten. The checklist is based on information from the 5 th edition of the ISO 9001:2015 and from the 3 rd edition of the ISO 13485:2016 International Standards for Quality Management Systems. While the ISO 13485:2016 standard is beneficial and very effective way to make document the questions that require to ask to make sure that the process outputs meet the planned arrangements for your process. Why to use audit checklists in internal audit? The audit checklist should cover questions on supplier evaluation, and an evolution of the supplier audit reports that have been collected, to see if they are done when examined by the QMS. The audit checklist is created by examining the ISO 13485:2016 standard and any documented procedures or undocumented processes for the activity to verify what happen. There are two sets of planned arrangements to check: which are required by ISO 13485:2016, and those that the company has put in place for their process to function.Īn ISO 13485 audit checklist is basically a set of questions that the auditor wants to ask, or activities that the auditor wants to observe, in order to verify the planned arrangements. How to create an audit checklist to check conformance?Īn ISO 13485 Audit is evidence to outcome of a process through a review of records of the employees, and then it is compared with the planned arrangements for the process to watch if what is being done is what was planned. So, when you are creating an ISO 13485 audit checklist, you want to include the information needed to ensure that you successfully check these two outcomes of the process. To ensure that the process is implemented and maintained effectively.To ensure that the processes are meeting the planned arrangements and regulatory requirements that the company has identified for the process in the QMS.As per clause 8.2.4 of the standard, the internal audit is to perform two functions: To better understand the why and how of internal audit checklists, it is beneficial to understand what the ISO 13485:2016 requirements determine about why we do internal audits. What are ISO 13485:2016 requirements for the audit? ISO 13485 Audit checklists are not revealed as an ISO 13485:2016 standard’s requirements, popularly they are a used and important tool to ensure that when you perform an internal audit on a process, as well as you make sure that just don’t miss any elements of that process. 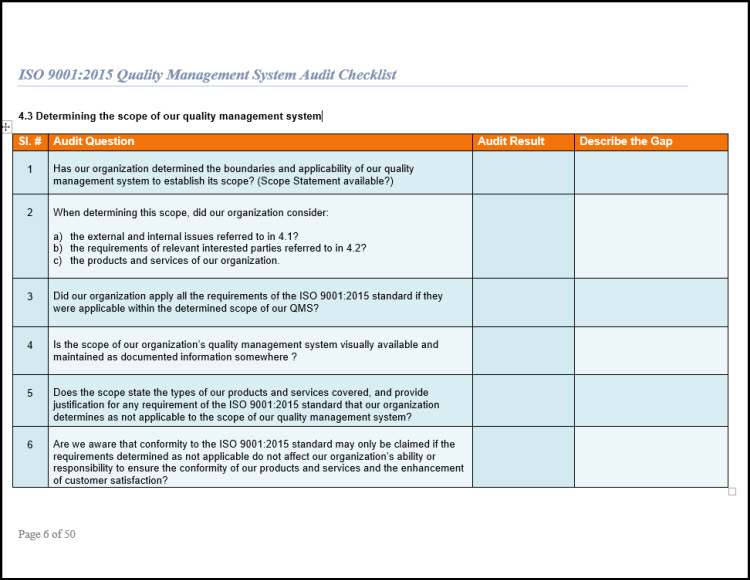
The ISO 13485:2016 requirements are transparent for all that it’s a critical element of your QMS and, since you want to know how your processes are performing, internal audits become a key resource. My company is small and low on the $$, and this will probably come out of my own pocket if I get it.One of the most important checking tools in a Quality Management System for medical devices, or any management system, is the internal audit. However, I wanted to see if anyone had used it and their opinion, before paying for it. 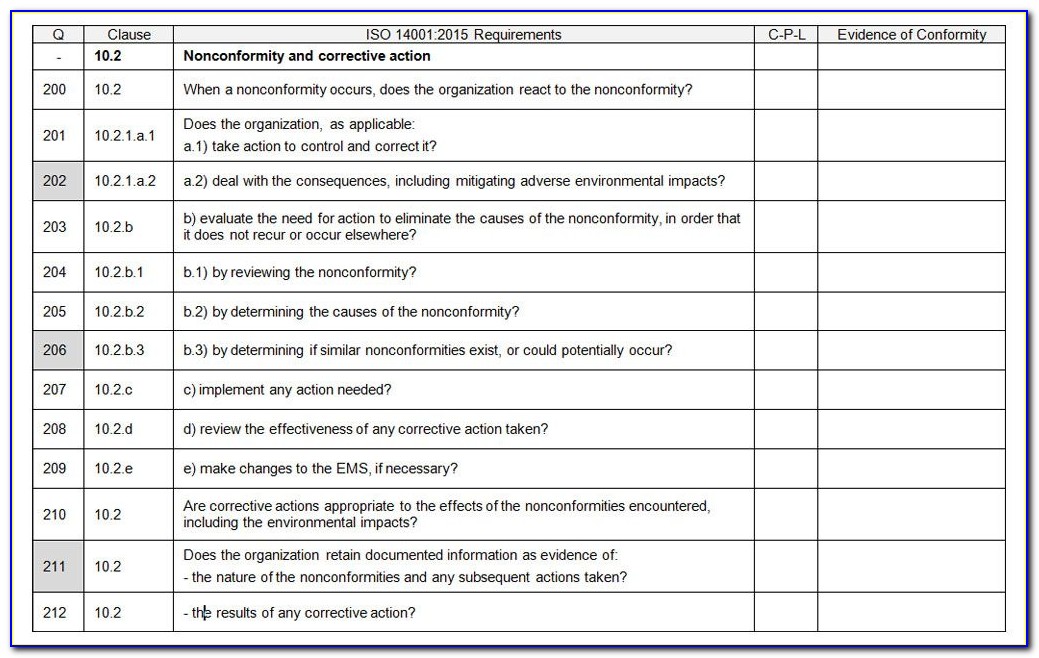
(I'd put the link in, but I haven't posted enough yet to do this).įrom the sample pages it looks great, with the right questions that will help me, as a novice, look for the right things during an audit, as well as be prepared for what external auditors will look for. ISO 13485 and FDA QSR (21 CFR 820) INTERNAL AUDIT CHECKLIST. This will tell you whether or not the processes are performing as planned, and if not, you. While stumbling around the Internet looking for help I came across this: After you have operated the QMS for the prescribed length of time, but before the certification body conducts their audit, you will need to perform an internal audit of each process. We've recently been audited and one finding was that the document we are using as a checklist/assessment tool does not cover ISO 13485, IVDD and FDA requriements (and we are subject to all of these). I've searched the forums for this topic, but couldn't find it so I apologize if it's already been discussed. I am new to the forum and new to the world of Quality, so please bear with me!
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
